Tissue Monitoring by Macrophages
Tissue Monitoring by Macrophages
Tissue Monitoring by Macrophages
Research Interests
Tissue resident macrophages are evolutionary old, tissue resident myeloid cells, found from invertebrates (e.g. D. melanogaster) to higher mammals (e.g. Homo sapiens). They are phagocytic cells and are generally characterised as tissue sentinels against invading pathogens. Yet, macrophages are far more than just bug-eaters! They actually have important roles in tissue homeostasis. We are generally interested to understand the functions of macrophages at steady state and in disease and how they regulate tissue homeostasis.
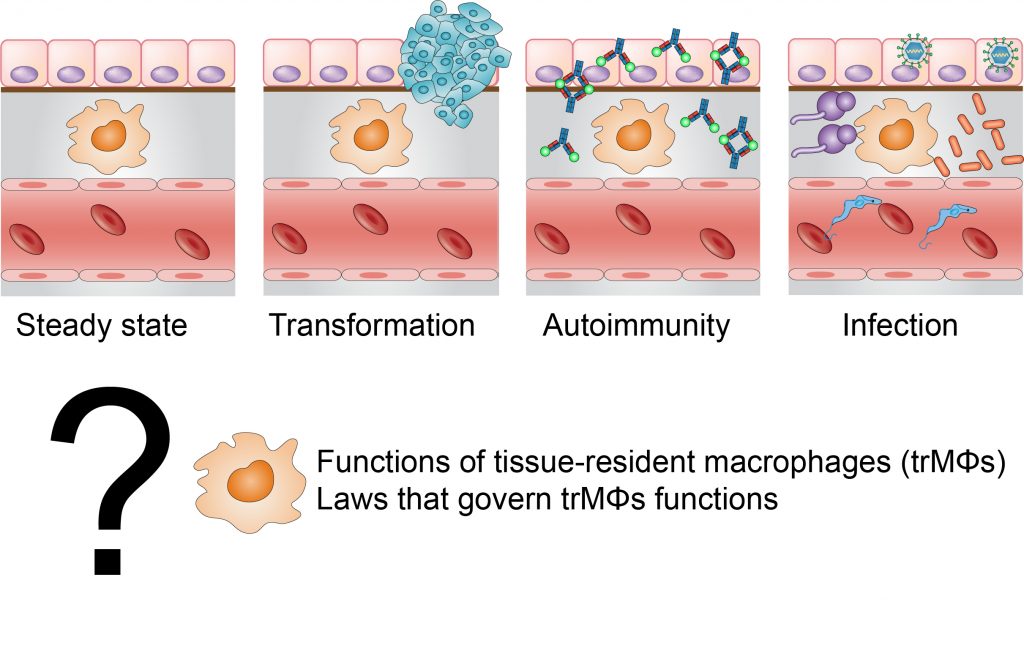
Elie Metchnikoff first described phagocytes (i.e. macrophages) more than a century ago in the star fish larvae as cells that fight against invading pathogens and as regulators of tissue homeostasis. Although, nowadays, the development of tissue resident macrophages has been extensively studied, their in situ functions are unclear and need to be better defined. In our endeavour to understand the role of macrophages within a specific tissue, we seek to answer six simple, yet, important, questions: Who? What? When? Where? Why? How?
Answering these fundamental questions will help us shed some light on the fascinating world of macrophages!

Research Methods
We employ a plethora of research techniques to study macrophages e.g. flow cytometry and gene expression. Since it is impossible to understand the biology of macrophages using cell lines or in vitro, we rely heavily on in vivo models. Importantly, we strongly believe that appropriate localisation of cell populations within their natural tissue setting is critical for relating cell diversity to function. Therefore, in order to answers our scientific questions, we are very keen on using state of the art imaging techniques: e.g. immunofluorescence (IF) microscopy, whole mount with tissue clearing, intravital microscopy, and transmission electron microscopy (TEM). If you are interested to see some representative pics and videos of our work, do have a look at our gallery!

Gallery – Seeing is believing
Snapshots
Movies
In vivo imaging of the renal cortex of a Cxc3r1gfp/+ mouse. The vasculature was labeled with i.v. injection of TRITC-Dextran. From Stamatiades et. al. Cell, 2016.
Probing of the tissue by macrophages (green) in the renal cortex of a Cxc3r1gfp/+ mouse. The vasculature was labeled with i.v. injection of TRITC-Dextran. From Stamatiades et. al. Cell, 2016.
Phagocytosis of beads by monocytes. From Carlin, Stamatiades et. al. Cell, 2013.
In vivo labeling of neutrophils. Carlin, Stamatiades et. al. Cell, 2013.
In vivo labeling of monocytes and neutrophils. Carlin, Stamatiades et. al. Cell, 2013.
Take up of immunce complexes (IC, red) by a kidney macrophage.
Uptake of ovalbumin in the liver.
3D rendering of a IC-loaded (red) kidney macrophage to demonstrate that IC are inside the macrophage.
Phagocytosis of M. marinum by a kidney macrophage. From Stamatiades et. al. Cell, 2016.
In vivo imaging of the glomerulus in a Cxc3r1gfp/+ mouse. The vasculature was labeled with i.v. injection of TRITC-Dextran.
In vivo imaging of a large blood vessel in the kidney of a Cxc3r1gfp/+ mouse. The vasculature was labeled with i.v. injection of TRITC-Dextran.
Phagocytosis of C. albicans by a kidney macrophage. From Stamatiades et. al. Cell, 2016.
Phagocytosis of 20 nm beads by a kidney macrophage. From Stamatiades et. al. Cell, 2016.
Knowledge sharing
Because science is all about sharing, we share any ‘expertise’ we have acquired and developed. To this, we regularly review online good antibodies for IF microscopy, in order to make IF less stressful for other colleagues: https://www.biocompare.com/General-Search/?search=stamatiades
Projects
The role of myeloid cells in immune complex-driven nephritis
Immune complexes (IC)-mediated pathologies (also known as type III hypersensitivity reactions) affect millions worldwide, with a very high socioeconomic burden. Type III hypersensitivity frequently targets the kidney, causing inflammation, which could progress to end stage renal failure and the need for dialysis or transplantation. The current therapeutic interventions for IC-mediated nephritis are costly, non-specific and come with severe side effects. A better understanding of the mechanistic underpinnings of the disease is needed, in order to build new, more specific treatments.
Myeloid cell infiltration is one of the most striking features of renal inflammation caused by IC and it correlates with poor patient prognosis. Our published work has shown that the macrophages that reside in the renal interstitium, termed kidney-resident macrophages (krMΦs), initiate type III hypersensitivity in the kidney, by scavenging circulating IC that are pumped into the renal interstitium by endothelial cells.
Our findings provide a simple model to explain how IC cause kidney inflammation. Yet, despite this progress, the underlying mechanisms that lead to IC-mediated tissue injury remain unclear. We aim to delineate the role of different subsets of myeloid cells in IC-driven inflammation in the kidney. Thus, our work has the potential to discover novel pathways that can be further harnessed for the prevention of type III hypersensitivity.

Contact us
If you have a passion about science, are interested in our work and would like to join us, get in touch! We are always looking for postdocs, students or interns. Send your CV and cover letter with your research interests to stamatiadeslab[at]gmail.com.
Publications (*equal contribution)
Kansler ER, Dadi S, Krishna C, Nixon BG, Stamatiades EG, Liu M, Kuo F, Zhang J, Zhang X, Capistrano K, Blum KA, Weiss K, Kedl RM, Cui G, Ikuta K, Chan TA, Leslie CS, Hakimi AA, Li MO (2022). Cytotoxic innate lymphoid cells sense cancer cell-expressed interleukin-15 to suppress human and murine malignancies. Nat Immunol. May 26. doi: 10.1038/s41590-022-01213-2. Epub ahead of print.
Chou C, Zhang X, Krishna C, Nixon BG, Dadi S, Capistrano KJ, Kansler ER, Steele M, Han J, Shyu A, Zhang J, Stamatiades EG, Liu M, Li S, Do MH, Edwards C, Kang DS, Chen CT, Wei IH, Pappou EP, Weiser MR, Garcia-Aguilar J, Smith JJ, Leslie CS, Li MO (2022). Programme of self-reactive innate-like T cell-mediated cancer immunity. Nature; 605:139-145.
Feng* Y, Dionne* MS, Stamatiades* EG, Kierdorf* K (2021). Deciphering phagocyte functions across different species. Front. Cell Dev. Biol; 9:712929.
Xu K, Yin N, Peng M, Stamatiades EG, Chhangawala S, Shyu A, Li P, Zhang X, Do MH, Capistrano KJ, Chou C, Leslie CS, Li MO (2021). Glycolytic ATP fuels phosphoinositide 3-kinase signaling to support effector T helper 17 cell responses. Immunity; 54:976-987.
Xu K, Yin N, Peng M, Stamatiades EG, Shyu A, Li P, Zhang X, Do MH, Wang Z, Capistrano KJ, Chou C, Levine AG, Rudensky AY, Li MO (2021). Glycolysis fuels phosphoinositide 3-kinase signaling to bolster T cell immunity. Science; 371:405-410.
Liu M, Kuo F, Capistrano KJ, Kang D, Nixon BG, Shi W, Chou C, Do MH, Stamatiades EG, Gao S, Li S, Chen Y, Hsieh JJ, Hakimi AA, Taniuchi I, Chan TA, Li MO (2020). TGF-β suppresses type 2 immunity to cancer. Nature; 587:115-120.
Li S, Liu M, Do MH, Chou C, Stamatiades EG, Nixon BG, Shi W, Zhang X, Li P, Gao S, Capistrano KJ, Xu H, Cheung NV, Li MO (2020). Cancer immunotherapy via targeted TGF-β signalling blockade in TH cells. Nature; 587:121-125.
Berrien-Elliott MM, Sun Y, Neal C, Ireland A, Trissal MC, Sullivan RP, Wagner JA, Leong JW, Wong P, Mah-Som AY, Wong TN, Schappe T, Keppel CR, Cortez VS, Stamatiades EG, Li MO, Colonna M, Link DC, French AR, Cooper MA, Wang WL, Boldin MP, Reddy P, Fehniger TA (2019). MicroRNA-142 Is Critical for the Homeostasis and Function of Type 1 Innate Lymphoid Cells. Immunity; 51:479-490.
Stamatiades EG, Li MO (2019). Tissue Resident Cytotoxic Innate Lymphoid Cells in Tumour Immunosurveillance. Seminars in Immunology; 41:101269
Stamatiades EG, Tremblay ME, Bohm M, Crozet L, Bisht K, Kao D, Coelho C, Fan Xi, Yewdell WT, Davidson A, Heeger PS, Diebold S, Nimmerjahn F, Geissmann F (2016). Immune Monitoring Of Trans-Endothelial Transport By Kidney Resident Macrophages. Cell; 166:991-1003.
Carlin* LM, Stamatiades* EG, Auffray C, Hanna RN, Glover L, Vizcay-Barrena G, Hedrick CC, Cook HT, Diebold S, Geissmann F (2013). Nr4a1-Dependent Ly6Clow Monocytes Monitor Endothelial Cells and Orchestrate Their Disposal. Cell; 153: 362-75.












